How the microbiota stimulates growth
|
|
- 1While insulin-like growth factor 1 (IGF-1), mainly produced by the liver, has a chemical structure similar to that of insulin, these hormones have distinct functions: IGF-1 stimulates tissue and skeletal growth, whereas insulin regulates energy metabolism as required for growth.
The gut microbiota is now thought to constitute an organ in its own right. The work of a team led by scientists from the CNRS and ENS de Lyon, in collaboration with Claude Bernard Lyon 1 University, INSERM, and INRAE, is the subject of an upcoming article in Science. These researchers have learned how a certain microbiotic bacterial strain stimulates the growth of undernourished young animals.
Though essential for health, microbiotic activity is still poorly understood. Through earlier studies, an international research team,1 directed by scientists from the Institute of Functional Genomics of Lyon (CNRS / ENS de Lyon), demonstrated that the gut microbiota plays a key role in the growth of juvenile individuals belonging to species as different as drosophilas and house mice.
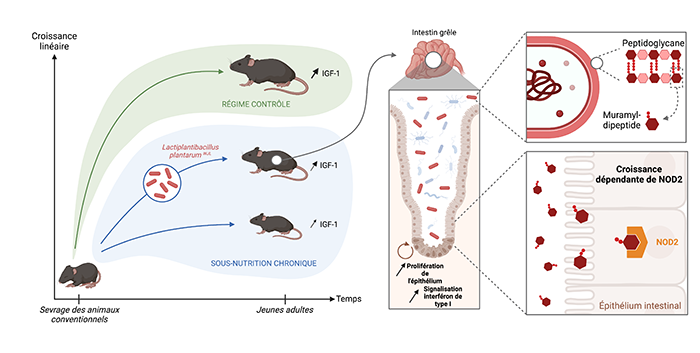
Specifically, one strain of the bacterium Lactobacillus plantarum (LpWJL) is very effective at stimulating growth in immature individuals under nutritionally deficient conditions. In this new study, the team identified one of the mechanisms by which LpWJL affects infant mice in a state of undernourishment after weaning.2
Daily oral administration of LpWJL to the young mice promotes maturation of the intestinal epithelium locally, which in turn bolsters the synthesis of hormones essential for healthy growth: insulin and IGF-1. The scientists determined that the bonding of muramyl dipeptide (a molecule produced by LpWJL that is a major component of the bacterial cell wall) to the NOD2 receptor on the intestinal epithelial cells of mice suffices to stimulate this hormone production.
The researchers’ findings thus demonstrate that muramyl dipeptide–NOD2 interaction helps lessen growth delays associated with chronic undernutrition. They suggest the potential of bacterial supplementation, in combination with other dietary measures, to enhance renewed growth in chronically undernourished children. Finally, they point to areas for further investigation concerning other populations that require optimized nutrition, including elderly individuals and high-level athletes.
© Amélie Joly
- 1Team members in France hailed from the Institute of Functional Genomics of Lyon; the Micalis Institute (INRAE / AgroParisTech / Paris-Saclay University); the Department of Paediatric Gastroenterology, Hepatology, and Nutrition at the Hospices Civils de Lyon; the Laboratory of Cell Physiology (INSERM / Lille University); and the Integrative and Molecular Microbiology (CNRS / Institut Pasteur) and CarMeN (INSERM / INRAE / Claude Bernard Lyon 1 University) research units. Their colleagues outside of France are affiliated with the Czech Academy of Sciences and the European Molecular Biology Laboratory (Germany).
- 2Weaning is a postnatal developmental phase marked by the end of suckling and the start of independent eating.
Microbe-mediated intestinal NOD2 stimulation improves linear growth of undernourished infant mice. Martin Schwarzer, Umesh Kumar Gautam, Kassem Makki, Anne Lambert, Tomas Brabec, Amelie Joly, Dagmar Šrůtková, Pierre Poinsot, Tereza Novotná, Stéphanie Geoffroy, Pascal Courtin, Petra Petr Hermanová, Renata C. Matos, Jonathan J.M. Landry, Céline Gérard, Anne-Laure Bulteau, Tomáš Hudcovic, Hana Kozaková, Dominik Filipp, Marie-Pierre Chapot-Chartier, Marek Šinkora, Noël Peretti, Ivo Gomperts Boneca, Mathias Chamaillard, Hubert Vidal, Filipe De Vadder, François Leulier, Science, 24 February 2023. doi: www.science.org/doi/10.1126/science.ade9767